One Root Cause, Four Diseases - How the Same Cellular Breakdown Drives Heart Disease, Cancer, Diabetes and Dementia
Most people are told heart disease, cancer, diabetes and dementia are separate, inevitable diseases. This article explains how one cellular ‘root‑cause triad’ drives them all – and how targeted nutrients like tocotrienols and geranylgeraniol can help you act early, protect your brain and body, and move from disease management to true prevention.
CANCERCARDIOVASCULAR HEALTHMETABOLIC HEALTHTYPE 2 DIABETESBRAIN HEALTHINFLAMMATIONBONE HEALTHCELLULAR HEALTH
Natural Health Connect Research Team
1/16/202611 min read
The modern healthcare system treats chronic diseases as if they were unrelated problems. One specialist manages your heart, another your blood sugar, another your bones, another your memory. Each adds another prescription, yet your underlying health keeps drifting in the wrong direction.
A very different picture has emerged from recent research. Heart disease, type 2 diabetes, cancer, dementia, osteoporosis and accelerated ageing are not separate “bad luck” events. They are different expressions of the same deep cellular breakdown.
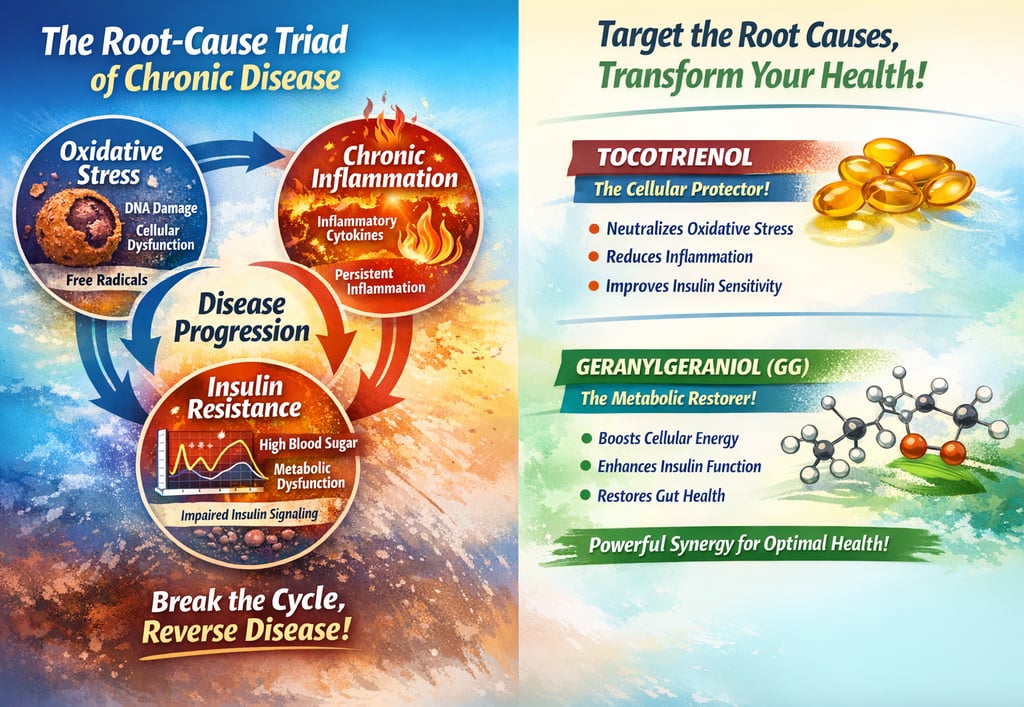
At the centre of this breakdown is a root‑cause triad:
Oxidative stress
Chronic inflammation
Insulin resistance
These three mechanisms are tightly interwoven and they damage every high‑energy organ in the body, including the brain. When you address them together, you are not treating four separate diseases, you are correcting one underlying process that expresses itself as heart disease, cancer, diabetes, dementia and more.
The Root‑Cause Triad - One Engine, Many Diseases
1. Oxidative stress – “rusting” of cells
Oxidative stress is an overload of reactive oxygen species (ROS) that damage DNA, proteins, lipids and mitochondria faster than your antioxidant defences can repair them. You can think of this as biological rust that slowly degrades blood vessels, pancreatic beta cells, neurons and bone tissue.
In arteries, oxidative stress oxidises LDL, injures the endothelium and accelerates plaque formation.
In cancer, it drives DNA damage and mutations.
In diabetes, it impairs insulin signalling and beta‑cell function.
In the brain, it injures synapses and accelerates neurodegeneration, particularly in Alzheimer’s disease.
2. Chronic inflammation – the smouldering fire
Acute inflammation heals injuries. Chronic, low‑grade inflammation is different, it’s a persistent, system‑wide “smoulder”, largely orchestrated by NF‑κB and pro‑inflammatory cytokines such as IL‑6 and TNF‑α.
This chronic fire:
Drives atherosclerosis and plaque instability in cardiovascular disease.
Creates a tumour‑promoting microenvironment and suppresses immune surveillance in cancer.
Impairs insulin signalling and worsens metabolic syndrome and type 2 diabetes.
Triggers microglial activation, neuroinflammation and progressive neuronal loss in dementia, including Alzheimer’s disease.
3. Insulin resistance – the metabolic disruptor
Insulin resistance is impaired cellular response to insulin, leading to hyperinsulinaemia, dysregulated PI3K/Akt/mTOR signalling, and disturbed glucose and lipid metabolism.
This has far‑reaching consequences:
Cardiovascular: accelerates atherosclerosis and endothelial dysfunction.
Cancer: hyperinsulinaemia and elevated IGF‑1 activate the PI3K/Akt pathway, promoting tumour growth, survival and angiogenesis.
Diabetes: leads to chronic hyperglycaemia and beta‑cell stress.
Brain: systemic and brain insulin resistance are now recognised as major risk factors for cognitive decline and Alzheimer’s disease, sometimes described as “type 3 diabetes”.
Dementia - The Missing Piece in the Chronic Disease Story
Dementia is often treated as a mysterious brain disease that appears late in life, unrelated to metabolic health. The evidence says otherwise.
Insulin resistance and the brain
Multiple human and mechanistic studies now show:
Peripheral insulin resistance and metabolic syndrome significantly increase the risk of Alzheimer’s and other dementias.
Insulin resistance is associated with reduced glucose uptake in key memory regions (hippocampus, temporal lobes) and with higher amyloid burden, even in middle‑aged adults at risk of Alzheimer’s.
Disrupted brain insulin signalling makes neurons more vulnerable to oxidative stress, mitochondrial dysfunction and inflammation, accelerating cognitive decline.
In other words, the same insulin resistance that drives diabetes and cardiovascular disease is silently reshaping the brain decades before dementia is diagnosed.
Oxidative stress and neuroinflammation
The brain is an energy‑intensive organ, consuming about 20% of the body’s energy. It is extremely sensitive to oxidative damage and inflammatory mediators.
Oxidative stress disrupts proteins involved in insulin signalling and damages mitochondria in neurons.
Chronic neuroinflammation, driven by NF‑κB and inflammatory cytokines, contributes to amyloid‑β accumulation, tau pathology and synaptic loss, hallmarks of Alzheimer’s disease.
Type 2 diabetes and repeated post‑meal glucose spikes further increase dementia risk, likely by worsening both oxidative stress and brain insulin resistance.
When you place dementia into this metabolic framework, it is not an outlier. It is one of the clearest examples of what happens when the root‑cause triad runs unchecked in the brain.
Why Drugs Alone Don’t Fix the Root Causes
Conventional medications can be lifesaving, but most are designed to blunt numbers and symptoms rather than repair the underlying cellular dysfunction.
Statins lower cholesterol, but they also deplete GGPP and CoQ10, which are crucial for mitochondrial function and insulin signalling. This can worsen insulin resistance and contribute to muscle symptoms.
Many diabetes drugs lower blood glucose without substantially improving chronic inflammation or oxidative stress.
Chemotherapy targets rapidly dividing cancer cells but generates intense oxidative stress and often leaves the pro‑cancer metabolic environment intact.
Bisphosphonates slow bone resorption but do not address why bone formation has declined, and may impair bone remodelling over time.
From a dementia perspective, this is even more stark, once neurodegeneration is established, symptom‑targeting drugs have modest effects at best. The most effective window is mid‑life prevention, precisely when insulin resistance, oxidative stress and chronic inflammation are already taking hold.
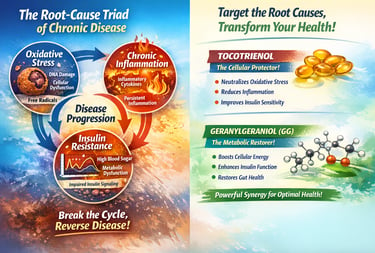
Two Annatto‑Derived Compounds that Act Upstream - Tocotrienol and Geranylgeraniol
Annatto seeds provide two natural compounds that act directly on the root‑cause triad and, importantly, have effects relevant to both systemic disease and brain health.
Tocotrienols – cellular and neurological protection
Tocotrienols are potent vitamin E isoforms with far stronger antioxidant and signalling effects than standard alpha‑tocopherol.
Key actions:
Oxidative stress: Tocotrienols neutralise ROS, protect cell membranes and support endogenous antioxidant systems.
Chronic inflammation: They inhibit NF‑κB activation and reduce inflammatory cytokines, helping cool the chronic inflammatory fire.
Insulin resistance: Tocotrienols modulate PPARs and protect insulin signalling components, improving insulin sensitivity in metabolic syndrome models and clinical studies.
Tocotrienols and dementia‑related mechanisms
Tocotrienols are one of the few lipid‑soluble antioxidants that can cross or reach the brain and have been shown to exert multiple neuroprotective effects:
Reduce amyloid‑related pathology and preserve cognitive performance in Alzheimer’s model animals.
Improve learning and memory in aged rats and in models of oxidative‑stress‑induced cognitive decline.
Suppress neuroinflammation and protect neurons via modulation of NF‑κB and other signalling pathways.
These findings align directly with the dementia arm of the root‑cause triad: oxidative stress, neuroinflammation and brain insulin resistance.
Geranylgeraniol (GG) – metabolic and mitochondrial restoration
Geranylgeraniol is a mevalonate‑pathway intermediate that replenishes geranylgeranyl pyrophosphate (GGPP), supporting protein prenylation, mitochondrial function and insulin signalling.
Key actions:
Supports endogenous CoQ10 synthesis, improving mitochondrial energy production.
Enhances insulin sensitivity through PPARγ activation and restoration of proper insulin receptor and GLUT4 function.
Modulates adipose inflammation and gut microbiota composition, contributing to systemic anti‑inflammatory and metabolic benefits.
From the perspective of brain health:
Better systemic insulin sensitivity and mitochondrial function reduce the metabolic overload that drives brain insulin resistance and neurodegeneration.
Improved vascular and metabolic health support cerebral blood flow and energy supply to neurons.
How the Triad Links Heart Disease, Cancer, Diabetes and Dementia
Here is how the same three mechanisms manifest as different diseases:
Oxidative stress
Heart & vessels: LDL oxidation, endothelial damage, plaque formation
Cancer: DNA damage, mutations, tumour progression
Diabetes & metabolic syndrome: Beta‑cell stress, impaired insulin signalling
Brain & dementia: Mitochondrial damage, synaptic loss, amyloid pathology
Chronic inflammation
Heart & vessels: Atherosclerosis, plaque instability
Cancer: Tumour microenvironment, immune evasion
Diabetes & metabolic syndrome: Worsened insulin resistance, NAFLD, obesity
Brain & dementia: Neuroinflammation, microglial activation, neuronal loss
Insulin resistance
Heart & vessels: Endothelial dysfunction, hypertension, dyslipidaemia
Cancer: Hyperinsulinaemia/IGF‑1 drive PI3K/Akt, tumour growth
Diabetes & metabolic syndrome: Hyperglycaemia, type 2 diabetes, metabolic syndrome
Brain & dementia: Reduced cerebral glucose uptake, cognitive decline, increased Alzheimer’s risk
When you intervene at the level of oxidative stress, chronic inflammation and insulin resistance, you are automatically acting on all four disease areas at once.
Where Tocotrienol + GG Fit In
Bringing it together:
Tocotrienols directly reduce oxidative stress and NF‑κB‑driven inflammation, improve insulin signalling, protect the cardiovascular system, modulate cancer‑related pathways and cross into the brain to exert neuroprotective effects.
Geranylgeraniol restores GGPP and CoQ10‑dependent mitochondrial function, improves insulin sensitivity, supports bone and muscle health, and counters statin‑induced muscle damage without blunting cholesterol‑lowering effects.
From a dementia‑inclusive perspective, this combination:
Addresses systemic insulin resistance that raises dementia and Alzheimer’s risk.
Reduces oxidative and inflammatory stress that drive neurodegeneration.
Supports brain energy metabolism through improved mitochondrial function and vascular health.
This is why pairing tocotrienol and geranylgeraniol is a rational strategy for whole‑body prevention: you are not “chasing” separate diseases; you are correcting the metabolic environment in which they arise.
From Disease Management to Prevention Medicine
Traditional care is largely reactive: wait until plaque is advanced, blood sugar is diabetic, tumours are visible, or memory is failing, then intervene. By that stage, the root‑cause triad has often been active for decades.
A prevention‑medicine perspective is different:
Identify and correct oxidative stress, chronic inflammation and insulin resistance early.
Support mitochondrial resilience and insulin signalling in both peripheral tissues and the brain.
Use targeted nutrients such as tocotrienols and geranylgeraniol, alongside lifestyle interventions, to normalise the underlying biology rather than just the lab numbers.
When you see heart disease, cancer, diabetes and dementia through this lens, they stop being four separate enemies. They become four warning lights on the same dashboard, all telling you it is time to fix the engine, not just reset the alerts.
Prevention medicine starts with understanding the root cause. Now you do.
References
Tocotrienol & Insulin Resistance
Chong JY, Huang TC, Chueh SM, et al. (2025). γ-Tocotrienol attenuates oxidative stress and preserves mitochondrial function in inflammation-induced muscle atrophy. Redox Biology, 87:103874.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12508907/
PMID: 41005207 | DOI: 10.1016/j.redox.2024.103874Rafique S, Khan DA, Farhat K, Noor M, Khan MA, Sharif M. (2023). Tocopherol Vs Tocotrienol (Vitamin E) in the Management of Metabolic Syndrome. Isra Medical Journal, 15(2):74-77.
https://www.imj.com.pk/wp-content/uploads/2024/02/RA-52-07-23.pdf
DOI: 10.55282/imj.ra52Mechanistic Insights into Antioxidant Interventions Targeting Obesity-Induced Oxidative Stress in the Pathogenesis and Complications of Type 2 Diabetes Mellitus (2025). Current Issues in Molecular Biology, 47(12):1063.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12731757/
PMC: PMC12731757
Geranylgeraniol & Insulin Sensitivity
Chung E, Elmassry MM, Cao JJ, Kaur G, Dufour JM, Shen CL. (2021). Beneficial effect of dietary geranylgeraniol on glucose homeostasis and bone microstructure in obese mice is associated with suppression of proinflammation and modification of gut microbiome. Nutrition Research, 93:27-37.
https://pubmed.ncbi.nlm.nih.gov/34352722/
PMID: 34352722 | DOI: 10.1016/j.nutres.2021.07.001Matsubara T, Takakura N, Urata M, et al. (2018). Geranylgeraniol Induces PPARγ Expression and Enhances the Biological Effects of a PPARγ Agonist in Adipocyte Lineage Cells. In Vivo, 32(6):1339-1344.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6365726/
PMID: 30348686 | DOI: 10.21873/invivo.11384
PI3K/Akt Pathway & Cancer
Fontana F, Giannitti G, Marchesi S, Limonta P. (2024). The PI3K/Akt Pathway and Glucose Metabolism: A Dangerous Liaison in Cancer. International Journal of Biological Sciences, 20(8):3113-3125.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11186371/
PMC: PMC11186371 | DOI: 10.7150/ijbs.89942Association of PTEN/PI3K/Akt pathway gene expression with insulin indices in adipose tissues of non-diabetic female adults: a cross-sectional study (2025). Nature Scientific Reports.
https://www.nature.com/articles/s41598-025-05233-4
DOI: 10.1038/s41598-025-05233-4Xingjun Huang et al. (2018). The PI3K/AKT pathway in obesity and type 2 diabetes. International Journal of Biological Sciences, 14(11):1483-1496.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6158718/
PMC: PMC6158718 | DOI: 10.7150/ijbs.24684Benjamin D Hopkins, et al. (2020). Insulin–PI3K signalling: an evolutionarily insulated metabolic driver of cancer. Nature Reviews Endocrinology, 2020 Mar 3;16(5):276–283.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7286536/
Tocotrienol & Cancer
Kjær IM, et al. (2023). Phase II trial of delta-tocotrienol in neoadjuvant breast cancer with evaluation of treatment response using ctDNA. Nature Scientific Reports, 13:8371.
https://www.nature.com/articles/s41598-023-35362-7
PMID: 37225860 | DOI: 10.1038/s41598-023-35362-7Ling MT, et al. (2012). Tocotrienol as a potential anticancer agent. Carcinogenesis, 33(2):233-239.
https://academic.oup.com/carcin/article/33/2/233/2463536
DOI: 10.1093/carcin/bgr265Younes M, et al. (2024). Review: Tocotrienol isoforms: The molecular mechanisms of their anticancer effects. Gene Reports, 34:101825.
https://www.sciencedirect.com/science/article/pii/S2095496424000025
DOI: 10.1016/j.genrep.2024.101825Naomi R, et al. (2021). An Interactive Review on the Role of Tocotrienols in the Modulation of Neurodegeneration. Frontiers in Nutrition, 8:754086.
https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2021.754086/full
DOI: 10.3389/fnut.2021.754086
NF-κB & Chronic Inflammation
Liu T, Zhang L, Joo D, Sun SC. (2017). NF-κB signaling in inflammation. Signal Transduction and Targeted Therapy, 2:e17023.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5661633/
PMC: PMC5661633 | DOI: 10.1038/sigtrans.2017.23Paul P Tak 1, Gary S Firestein (2001). NF-κB: a key role in inflammatory diseases. Journal of Clinical Investigation, 107(1):7-11.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC198552/
PMC: PMC198552 | DOI: 10.1172/JCI11830YS S Makarov (2000). NF-kappaB as a therapeutic target in chronic inflammation: recent advances. Mol Med Today. 2000 Nov;6(11):441-8.
https://www.sciencedirect.com/science/article/abs/pii/S0165614700018141Lawrence T. (2009). The nuclear factor NF-κB pathway in inflammation. Cold Spring Harbor Perspectives in Biology, 1(6):a001651.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2882124/Shin Yii Ang et al., Modulation of NF-κB signaling pathway by tocotrienol in neuroinflammation (2025). Discov Ment Health. 2025 Oct 24;5(1):160
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12552194/
PMC: PMC12552194
Tocotrienol Neuroprotection & Blood-Brain Barrier
Fukui K. (2019). Neuroprotective and Anti-Obesity Effects of Tocotrienols. Journal of Nutritional Science and Vitaminology, 65(Supplement):S185-S192.
https://www.jstage.jst.go.jp/article/jnsv/65/Supplement/65_S185/_pdf
DOI: 10.3177/jnsv.65.S185Kumari M, et al. (2021). Tocotrienols Ameliorate Neurodegeneration and Motor Deficits in the 6-OHDA-Induced Rat Model of Parkinsonism: Behavioural and Immunohistochemistry Analysis Nutrients
. 2021 May 10;13(5):1583.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8150907Angela Maria Mathew et al., Exploring the anti-inflammatory activities, mechanism, and neuroprotective effects of tocotrienols (2023). F1000Res 2023 Mar 27;12:338.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11406131/
PMC: PMC11406131
Geranylgeraniol & Statin Muscle Damage
Tan B, et al. (2023). Potential role of geranylgeraniol in managing statin-associated muscle symptoms: a systematic review. Frontiers in Physiology, 14:1202485.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10691100/
PMC: PMC10691100 | DOI: 10.3389/fphar.2023.1202485Irwin JC, et al. (2020). Geranylgeraniol prevents statin-induced skeletal muscle fatigue without blocking reductions in circulating cholesterol. Journal of Cachexia, Sarcopenia and Muscle, 11(5):1174-1186.
https://pubmed.ncbi.nlm.nih.gov/31491372/
PMID: 31491372 | DOI: 10.1002/jcsm.12575Jaśkiewicz A, et al. (2018). Geranylgeraniol Prevents Statin-Dependent Myotoxicity. Nutrients, 10(7):899.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5987243/
PMC: PMC5987243 | DOI: 10.3390/nu10070899Cao P, et al. (2009). Statin-induced muscle damage and atrogin-1 induction is the result of a geranylgeranylation defect. FASEB Journal, 23(11):3746-3757.
https://faseb.onlinelibrary.wiley.com/doi/full/10.1096/fj.08-128843
DOI: 10.1096/fj.08-128843Barajas B, et al. (2022). Geranylgeranyl pyrophosphate depletion by statins compromises skeletal muscle insulin sensitivity. Journal of Cachexia, Sarcopenia and Muscle, 13(3):1646-1660.
https://onlinelibrary.wiley.com/doi/full/10.1002/jcsm.13061
Tocotrienol & Bone Health
Maureen L. Meister, et al., (2020). Tocotrienols in Bone Protection: Evidence from Preclinical Studies. efood.k.200427.001
https://onlinelibrary.wiley.com/doi/full/10.2991/efood.k.200427.001
DOI: 10.2991/efood.k.200427.001Chin KY, Ima-Nirwana S. (2015). The biological effects of tocotrienol on bone: a review on evidence from rodent models. Drug Design, Development and Therapy, 9:2471-2482.
https://www.dovepress.com/the-biological-effects-of-tocotrienol-on-bone-a-review-on-evidence-fro-peer-reviewed-fulltext-article-DDDT
PMC: PMC4414160Nur Fathiah Mohd Radzi, Noor Akmal Shareela Ismail , Ekram Alias (2018). Tocotrienols Regulate Bone Loss through Suppression on Osteoclast Differentiation and Activity: A Systematic Review. Current Drug Targets, 19(9):1095-1107.
https://pubmed.ncbi.nlm.nih.gov/29412105/
PMID: 29412105
Insulin Resistance, Inflammation & Cancer Connection
Xin Zheng, et al., Temporal relationship between chronic inflammation and insulin resistance and their combined cumulative effect on cancer risk: a longitudinal cohort study (2025). BMC Medicine, 23:162.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12016061/
PMC: PMC12016061 | DOI: 10.1186/s12916-025-03695-zGuo-Tian Ruan, et al. (2022). Prognostic importance of systemic inflammation and insulin resistance in patients with cancer: a prospective multicenter study. BMC Cancer. 22:700.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9233357/
PMC: PMC9233357 | DOI: 10.3390/nu14122457Biagio Arcidiacono edt al., (2011). Insulin resistance and cancer risk: an overview of the pathogenesis. Exp Diabetes Res. 2012:789174.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3372318/Emily Jane Gallagher, Derek LeRoith (2010). The Proliferating Role of Insulin and Insulin-Like Growth Trends Endocrinol Metab., 21(10):610–618..
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2949481/Pollak M. (2008). Insulin and insulin-like growth factor signalling in neoplasia. Nature Reviews Cancer, 8:915-928.
https://www.nature.com/articles/nrc2536
DOI: 10.1038/nrc2536
Additional Supporting Evidence
Looi AD, et al. (2025). Health Benefits of Palm Tocotrienol-Rich Fraction: A Scoping Review. Nutrition Reviews, 83(2):307-326.
https://academic.oup.com/nutritionreviews/article/83/2/307/7698337
DOI: 10.1093/nutrit/nuae161Pulliero A, et al. (2025). Antioxidant Food Supplementation in Cancer: Molecular Mechanisms and Clinical Evidence. International Journal of Molecular Sciences, 26(19):10234.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12561932/
PMC: PMC12561932 | DOI: 10.3390/ijms261910234Mohamed Shafras, et al., Role of dietary antioxidants in diabetes: An overview (2024). Food Chemistry: X, https://www.sciencedirect.com/science/article/pii/S2772753X24000625#:~:text=According%20to%20research%2C%20antioxidant%20therapy%20protects%20the,natural%20dietary%20antioxidants%20in%20mitigating%20that%20effect.
Disclaimer
This article is for educational purposes only. The information presented is based on scientific research but should not replace professional medical advice. Always consult with qualified healthcare providers before making changes to your health regimen, especially if you have existing medical conditions or are taking medications.
For more evidence-based health information and to explore Natural Health Connect's research-backed supplements, visit www.naturalhealthconnect.com.au
